What is Galvanising?
We have all seen plenty of galvanised products, the textured silver surface appearance is instantly recognisable and seen on everything from joist hangers to motorway barriers, its applications are nearly endless!

However, have you ever wondered what is galvanising? No, well maybe it is time you did as not all galvanised products are of the same quality.
Galvanisation is the process of applying a zinc coat to another metal (typically steel). This process isn’t particularly new and examples of zinc coating date back as far as the 17th century, but modern methods have come a long way in the last 400 years.
Modern galvanising is typically achieved through a process called hot-dip galvanisation, here metallic items (typically steel parts) are dropped into superheated molten baths of zinc. Upon contact the zinc ‘clings’ to the surface and creates a perfect coat across the metal’s surface, covering it with an incredibly thin but highly resistive finish.
How Does Galvanisation Work?
Zinc on its own has limited use beyond health supplements and batteries, but when coated to another metal it has brilliant anti-corrosion properties. Mainly it is incredibly resilient to oxidisation (rust)!
To understand how galvanisation works, first you need to know what the reactivity series is. No, it isn’t a new Netflix sci-fi show, the reactivity series is a table of metal elements. The nearer the top of the table the more reactive the metal is. For example, potassium at the top of the table will explode when dropped in water! While platinum, at the bottom, will never rust (oxidise).
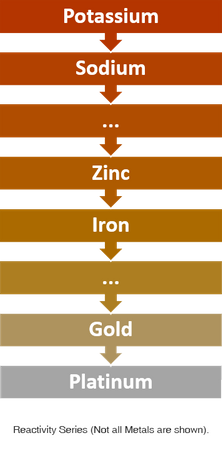
The series is important as it tells us how metals will behave when they are combined. When in a corrosive environment like a humid room or sea water a metal that is higher up the table will ‘sacrifice’ itself for a metal that is lower down the table.
This is fundamentally how a galvanisation works, the zinc coating acts as a barrier and corrodes on behalf of the steel (Iron) it is protecting. And as zinc takes longer to corrode than steel, galvanised products can withstand corrosive internal and external environments for a far greater length of time. Plus, the thicker the coating of zinc the longer it will protect from corrosion! Zinc corrosion is also a lot less noticeable than steel ‘rust’, so zinc products look great for longer than un-plated items.
Is all Galvanisation the Same?
Wouldn’t it be nice if it was that simple but unfortunately it isn’t! Hot-dip galvanisation is set out by British Standard BS EN 10346:2009 and includes Z100, Z275 and Z600 coatings.
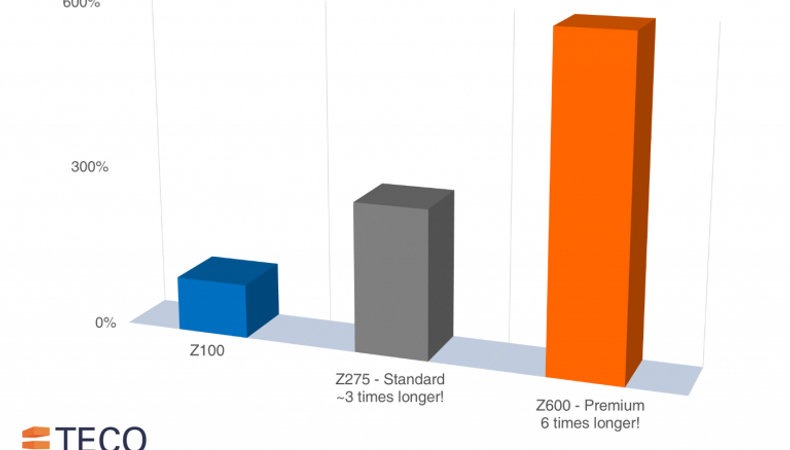
Each coating is a set thickness and engineered for specific applications, Z100 is the thinnest coverage at 7µm (Micrometres) and is designed for indoor applications only. Z600 is the thickest at 42µm (Micrometres) and provides premium protection against corrosion. The numbering system works like sun factor ratings, with a higher number signifying more protection!
For the building and engineering industry Z275 is the expected standard, this coating is 20µm (Micrometres) thick meaning the steel underneath has nearly three times as much zinc coverage when compared to Z100 coated items.
But what does this mean for your products? Well it depends on where you live but fortunately BS EN standards make it easier to work out using set corrosion categories. These categories range from C1 to C5 with C1 being the least corrosive environment and C5 being the piranha of corrosive environments as it will happily rust steel in no time at all! In the UK most places fall into category C2 or C3, some highland areas can be C4. This means that anywhere between 0.1 to 2.1 µm (Micrometres) of zinc coating will lose its effectiveness each year.
How Long Does Galvanisation Last?
For products such as TECO Single Piece Masonry Hangers (Z600 coated) in a C3 environment typically you can expect a minimum corrosion protection of 30 years but this could be as long as 60 years! For an inferior Z100 product it may only last around five years before the steel beneath the galvanised coating starts to rust (If the product is in contact with the ground visible oxidisation may set in even earlier!).
To find out the corrosion level in your area use the UK corrosion map created by the UK Galvanizers Association https://www.galvanizing.org.uk/corrosion-map/
Challenge Your Suppliers!
At TECO we pride ourselves on using Z275 galvanisation as a minimum with many of our products using Z600 coatings to give exceptional protection against corrosion (Approximately six times as much protection when compared to Z100!).

Many other products on sale today use inferior Z100 galvanisation and market them for unsuitable environments. This can lead to the item oxidising quickly or even failing as rust takes hold of the steel beneath!
You wouldn’t use sun factor 15 suncream in the Sahara desert when better protections exists. So why would you use Z100 galvanised products instead of superior Z275 and Z600 products?
We encourage you to look for Z275 or Z600 coatings in an item’s specification when purchasing galvanised products, if it isn’t there ask the supplier.
A good job deserves quality products so don’t let inferior galvanised goods reflect poorly upon your work!
Within the UK there is a representative body for galvanising manufacturers, these dedicated individuals have spent years researching and guiding the UK industry. For more information on galvanisation, please visit their website at https://www.galvanizing.org.uk/
